老化相關心血管疾病研究團隊
老化相關心血管疾病研究團隊
|
|
|
|
主持人: 王鐘賢 教授 |
研究專長:心肺物理治療、運動生理、血栓/發炎免疫
研究團隊成員:
王鐘賢老師、趙崇義老師、馬蘊華老師、楊春茂老師、周淑娥老師、陳喬男老師、傅鐵城醫師、黃書群醫師、王兆宏醫師
研究架構圖:
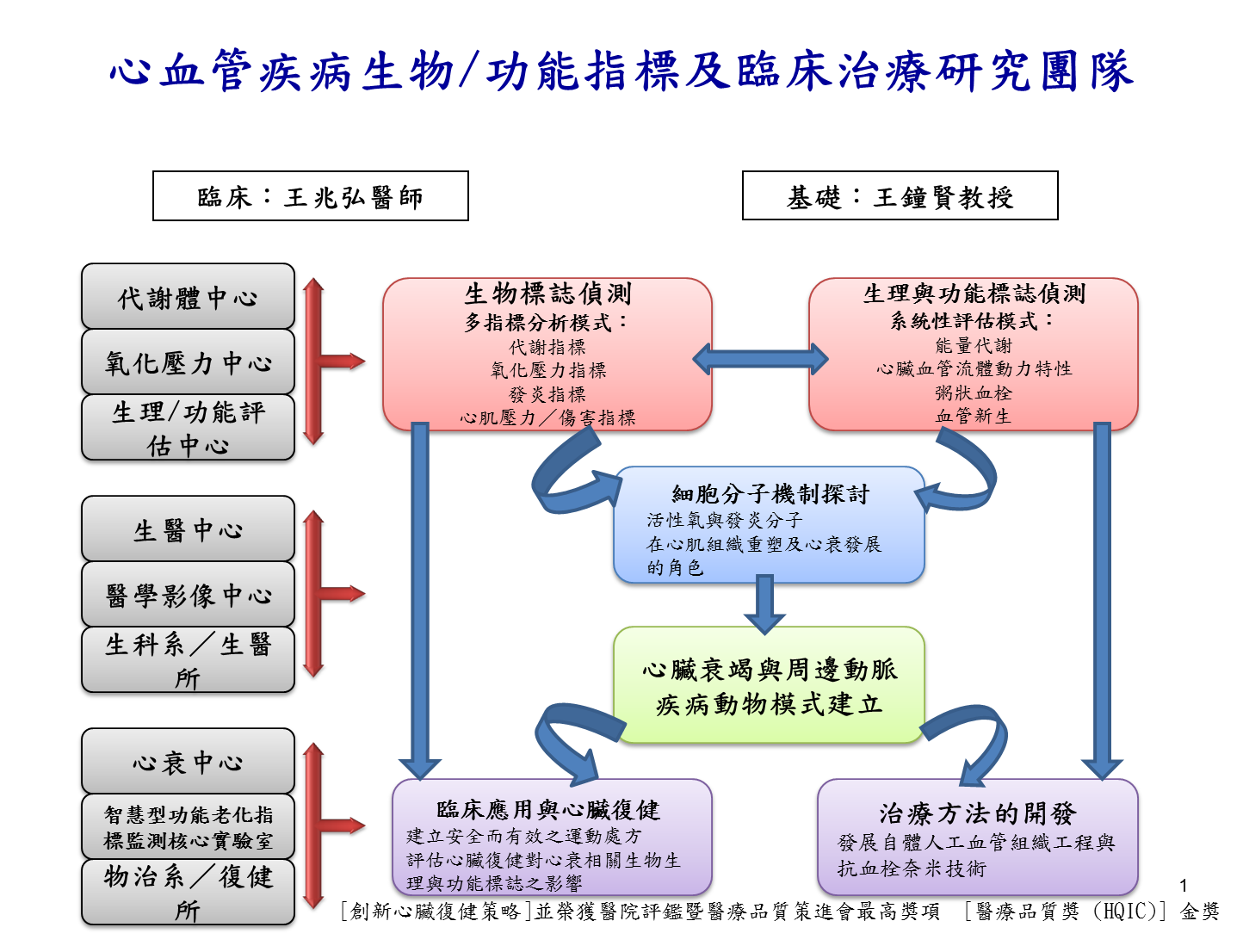
研究計畫:
說明: 本研究團隊擬以多指標分析模式建立生物偵測標誌,如生理/功能標誌;探討心肌組織重塑及心衰發展之分子機制及評估心臟復健對心衰相關標誌的影響,最終建立安全而有效的運動處方。
背景: 自民國96年起,心臟病已攀升至國人主要死亡原因之第二名,且死亡人數逐年增加。心臟衰竭是許多心臟疾病的病程終點,而在發病案例中有超過80%為高齡者。並且,罹患嚴重心臟衰竭之病人(NYHA功能分級IV),平均年死亡率更高達40~50%。研究調查顯示,由於人口老化、老年人口比例提高、以及冠狀動脈疾病存活的病人數日益增加等原因,加劇了心臟衰竭病患增加的趨勢。因此,降低心臟衰竭的發生與減緩心臟衰竭病生理進程的速度,是現今乃至未來極為重要的老化醫療議題。心臟衰竭患者可能因長期的神經內分泌代償失衡,導致心肌功能更加衰弱惡化,並引發末端器官和骨骼肌缺血、發炎、與機能損傷,使其病患日常活動能力與生活品質更加降低。然而,有關心臟衰竭確切的病生理機轉,目前尚未完全瞭解,亟待進一步研究釐清。由於心臟病變初期不易察覺,待病症出現時,常已邁入心臟衰竭階段。因此,開發偵測早期心臟病變及爾後衍生心臟衰竭的生物與功能指標,對降低心臟衰竭的發生與改善心臟衰竭病程發展之臨床決策判斷,極其重要。
目的: 本研究團隊擬以多指標分析模式建立生物偵測標誌,如生理/功能標誌;探討心肌組織重塑及心衰發展之分子機制及評估心臟復健對心衰相關標誌的影響,最終建立安全而有效的運動處方。
方法: 以系統性評估模式建立心臟衰竭[生理與功能標誌]偵測方法,建立心臟血管流體動力特性、粥狀血栓、血管新生、能量代謝等指標。
主要成果:
1. FU, Tieh-cheng, et al. Aerobic interval training improves oxygen uptake efficiency by enhancing cerebral and muscular hemodynamics in patients with heart failure. International journal of cardiology, 2013, 167.1: 41-50.
2. WANG, Jong-Shyan, et al. Exertional periodic breathing potentiates erythrocyte rheological dysfunction by elevating pro-inflammatory status in patients with anemic heart failure. International journal of cardiology, 2013, 167.4: 1289-1297.
3. WANG, Jong-Shyan, et al. Effect of aerobic interval training on erythrocyte rheological and hemodynamic functions in heart failure patients with anemia.International journal of cardiology, 2013, 168.2: 1243-1250.
4. CHEN, Yu-Wen, et al. Anemic comorbidity reduces capacity of endogenous thrombin generation and is associated with consumptive coagulopathy in patients with heart failure. International journal of cardiology, 2013, 168.5: 4965-4967.
5. CHEN, Yu-Wen; CHEN, Yi-Ching; WANG, Jong-Shyan. Absolute hypoxic exercise training enhances in vitro thrombin generation by increasing procoagulant platelet-derived microparticles under high shear stress in sedentary men. Clinical Science, 2013, 124.10: 639-649.
6. LEE, I.-Ta, et al. ATP stimulates PGE 2/cyclin D1-dependent VSMCs proliferation via STAT3 activation: role of PKCs-dependent NADPH oxidase/ROS generation. Biochemical pharmacology, 2013, 85.7: 954-964.
7. WENG, Tzu-Pin, et al. Effects of interval and continuous exercise training on CD4 lymphocyte apoptotic and autophagic responses to hypoxic stress in sedentary men. PloS one, 2013, 8.11: e80248.
8. HUANG, S. C., et al. Systemic vascular resistance is increased and associated with accelerated arterial stiffening change in patients with chronic cervical spinal cord injury. European journal of physical and rehabilitation medicine, 2013, 49.1: 41-49.
9. FU, Tieh-Cheng, et al. Cardiac Rehabilitation in Patients with Heart Failure.Acta Cardiologica Sinica, 2014, 30.5: 353-359.
10. WANG, Jong-Shyan, et al. Hypoxic exercise training improves cardiac/muscular hemodynamics and is associated with modulated circulating progenitor cells in sedentary men. International journal of cardiology, 2014, 170.3: 315-323.
11. WENG, Tzu-Pin, et al. Activation of lymphocyte autophagy/apoptosis reflects haemodynamic inefficiency and functional aerobic impairment in patients with heart failure. Clinical Science, 2014, 127.10: 589-602.
12. CHOW, Shu-Er, et al. Resveratrol induced ER expansion and ER caspase-mediated apoptosis in human nasopharyngeal carcinoma cells. Apoptosis, 2014, 19.3: 527-541.
13. HUANG, Shu-Chun, et al. Comparison of Cardiac Autonomic Nervous System Disturbed by Sleep Deprivation in Sex and Menstrual Phase. The Chinese journal of physiology, 2015, 58.2.
14. WANG, Jong-Shyan, et al. Effects of normoxic and hypoxic exercise regimens on monocyte-mediated thrombin generation in sedentary men. Clinical Science, 2015, 129.4: 363-374.
15. FU, Tieh-Cheng, et al. Aerobic Interval Training Elicits Different Hemodynamic Adaptations Between Heart Failure Patients with Preserved and Reduced Ejection Fraction. American Journal of Physical Medicine & Rehabilitation, 2016, 95.1: 15-27.
16. CHEN, Yi-Ching, et al. Interval and continuous exercise regimens suppress neutrophil-derived microparticle formation and neutrophil-promoted thrombin generation under hypoxic stress. Clinical Science, 2015, 128.7: 425-436.
17. TU, Shu-Ju, et al. Retention assessment of magnetic nanoparticles in rat arteries with micro-computed tomography. Physics in medicine and biology, 2014, 59.5: 1271.
18. LU, Yi-Ching, et al. Augmented cellular uptake of nanoparticles using tea catechins: effect of surface modification on nanoparticle–cell interaction.Nanoscale, 2014, 6.17: 10297-10306.
19. HO, Hung-Yao, et al. Characterization of global metabolic responses of glucose-6-phosphate dehydrogenase-deficient hepatoma cells to diamide-induced oxidative stress. Free Radical Biology and Medicine, 2013, 54: 71-84.
20. YANG, H. C., et al. Glucose 6-phosphate dehydrogenase deficiency enhances germ cell apoptosis and causes defective embryogenesis in Caenorhabditis elegans. Cell death & disease, 2013, 4.5: e616.
21. LIN, Hsin-Ru, et al. Proteome-wide dysregulation by glucose-6-phosphate dehydrogenase (G6PD) reveals a novel protective role for G6PD in aflatoxin B1-mediated cytotoxicity. Journal of proteome research, 2013, 12.7: 3434-3448.
22. CHENG, M. L., et al. Effective NET formation in neutrophils from individuals with G6PD Taiwan-Hakka is associated with enhanced NADP+ biosynthesis.Free radical research, 2013, 47.9: 699-709.
23. HO, H.-Y.; CHENG, M.-L.; CHIU, DT-Y. Glucose-6-phosphate dehydrogenase–beyond the realm of red cell biology. Free radical research, 2014, 48.9: 1028-1048.
24. TANG, Hsiang-yu, et al. Inability to maintain GSH pool in G6PD-deficient red cells causes futile AMPK activation and irreversible metabolic disturbance.Antioxidants & redox signaling, 2015, 22.9: 744-759.
25. YANG, Hung-Chi, et al. Glucose 6-phosphate dehydrogenase knockdown enhances IL-8 expression in HepG2 cells via oxidative stress and NF-κB signaling pathway. Journal of Inflammation, 2015, 12.1: 1.
26. WU, Yi-Hsuan, et al. Glucose-6-Phosphate Dehydrogenase Enhances Antiviral Response through Downregulation of NADPH Sensor HSCARG and Upregulation of NF-κB Signaling. Viruses, 2015, 7.12: 6689-6706.
27. CHEN, Chiao-Nan, et al. Home-‐Based Exercise May Not Decrease the Insulin Resistance in Individuals With Metabolic Syndrome. Journal of physical activity & health, 2015, 12.1: 74-79.
【獲獎】首創系統性及時評估運動心臟動力學變化與大腦/活動骨骼肌血流運行與氧氣利用的效率,榮獲「國家品質標章 (SNQ)」及2012年財團法人醫院評鑑暨醫療品質策進會最高獎項「醫療品質獎(HQIC)金獎」
【專利】王鐘賢: 智慧型腳踏車及操作方法
中華民國發明專利第I458521號(2012/10/19)
聯絡窗口: 楊玫齡 小姐 03-2118800# 3173
johfjk@mail.cgu.edu.tw








